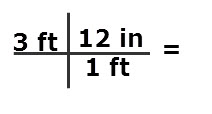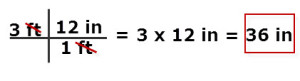A mole is a chemical unit of measure.
Dimensional analysis is a method used to convert one unit of measurement to another. Conversion factors are mathematical tools for converting between units of measurement. The conversion factor 1 ft = 12 inches is used to convert units of feet to units of inches or vise versa.
Example: How many inches are in 3 feet?
Think:
- What conversion factor compares the units of feet and inches?
Conversion factors are ratios between units. 1 ft :12 in; 1 ft/12 in - In the dimensional analysis method, conversion factors are written as fraction.
1 ft/12 in or 12 in/1 ft - Start with the measurement to be converted, which is 3 ft in the problem.
Write this measurement and draw two lines as shown. The horizontal line separates the numerator from the denominator. The vertical line separates factors that are multiplies.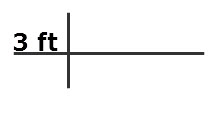
- Next, select the correct version of the conversion factor the converts the unit of feet to the unit of inches.
- Conversion factors are fractions. Remember when working with fractions, the first thing is to simply them: Do this by cancelling factors which are in common between the numerator and denominator. Factors can be numbers and or units of measurement.
- In order to cancel the foot unit, the conversion factor fraction used is 12 in/1 ft.
This is because the foot unit is in the denominator, thus the foot units cancel out as shown. - Remember that the vertical line separates fractions that multiplied.
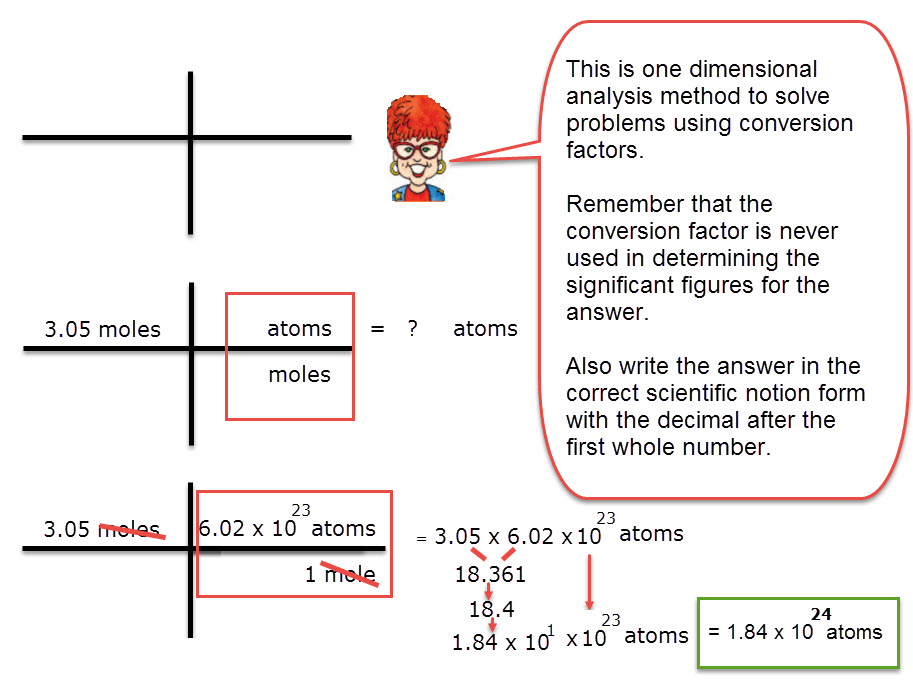
Problem 1: Use Avogadro’s Number (a conversion factor) to decide how many atoms are in 3.05 moles of calcium, Ca.
Clues:
- Write the amount that is to be converted.
- Add two lines, one to separate the numerators from the denominators. The second vertical line separates fractions that are multiplied.
- Add the conversion factor.
- Cancel units.
- Multiply factors in the numerator. Divide by factors in the denominator.
Check your work with the following:
Problem 2: How many moles lithium, Li are equal to 4.2 x 10 26 atoms of lithium?
Click How Many Atoms in a Mole? to find the answer for this problem.